
Assaying Tetracyclines by HPLC
Using the Allure' Biphenyl Stationary Phase
By Rick Lake,Pharmaceutical I nnovations Chemist, and Sherry Moyer, Innovations Chemist
• Superior selectivity and efficiency, using an Allure' Biphenyl column.
• Simplified analysis fo r high-throughput potency and stability-indicating assays.
• More easily achievable system suitab ility criter ia.
Tetracyclines are a widely used class of ant ibiotics
whose applications range from topical acne med ications for humans to premix feed additives for
livestock. Because of their widespread and liberal
use, tetracyclines are manufactured in large qua n
tities, which generates th e need for a large number
of potency and stability-indicating assays. These
assays must be comp leted at regular intervals, in a
timely manner, over extended periods of time.
Consequently, it is critical that simple, rugged, and
selective met hods be developed. By selecting a sta
tionary phase that produces optimum selectivity,
less demand to prod uce selectivity is placed on the
mobile phase, and a simp le isocratic analysis is
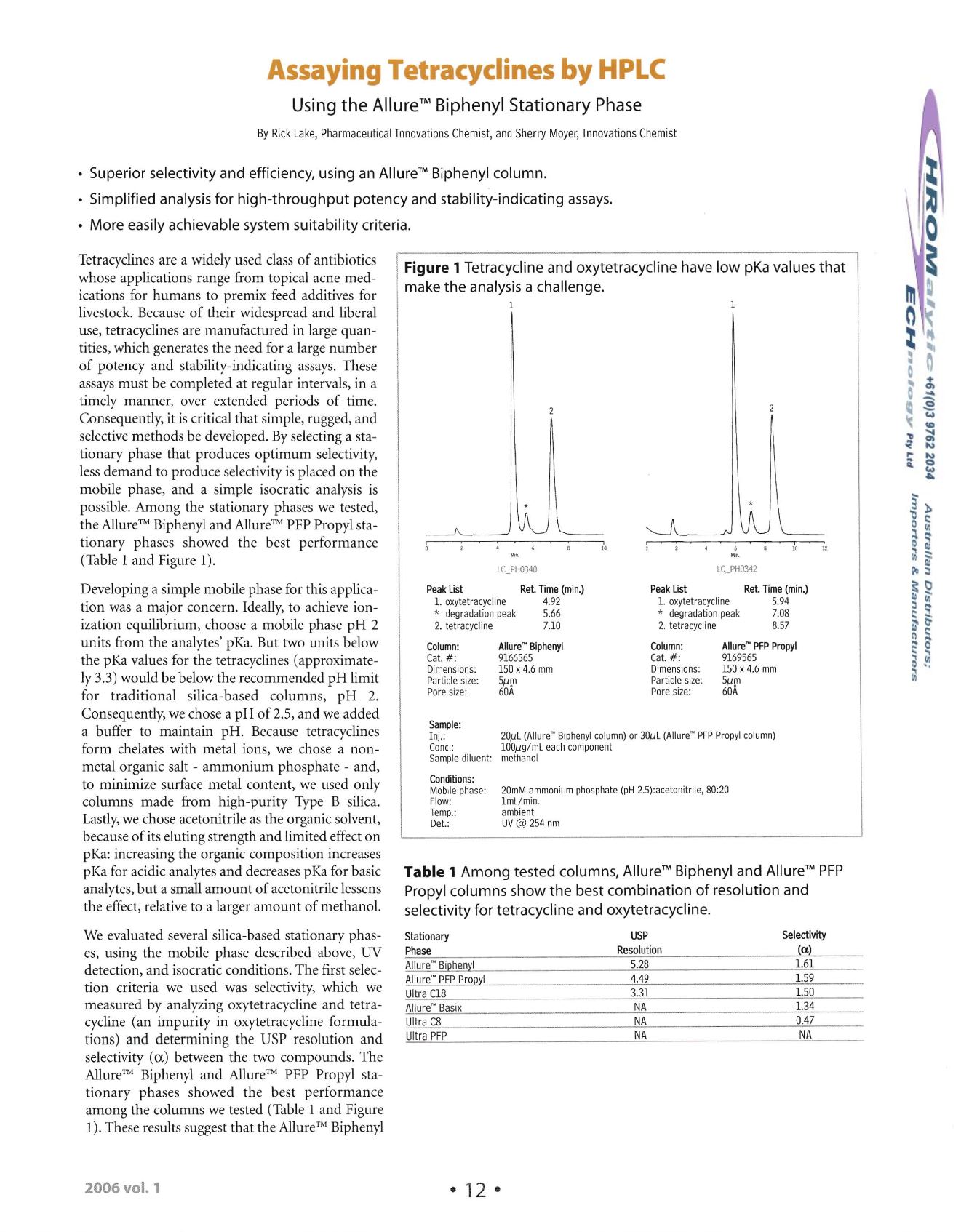
possible. Among the stationary phases we tested,
the Allure" Biphenyl and Allure" PFP Propyl sta
tionary ph ases showed the best performance
(Table 1 and Figure
1).
Developing a simple mobile phase for this applica
tion was a major concern . Ideally, to achieve ion
ization equilibri um, choose a mob ile phase pH 2
units from the analytes' pKa. But two units below
the pKa values for the tetracyclines (approximate
ly 3.3) would be below the recommended pH limit
for traditional silica-based columns, pH 2.
Consequently, we chose a pH of 2.5, and we added
a buffer to maintain pH. Because tetracyclines
form chelates with metal ions, we chose a non
metal organic salt - ammonium phosphate - and,
to minimize surface metal content, we used only
columns made from high-pur ity Type B silica.
Lastly,we chose aceton itrile as the organic solvent,
because of its eluting strengt h and limited effect on
pKa: increasing the organic composition increases
pKa for acidic analytes and decreases pKa for basic
analytes, bu t a small amount of acetoni trile lessens
the effect, relative to a larger amount of methanol.
We evaluated several silica-based statio nary phas
es, using the mobile phase described above, UV
detection, and isocratic conditions . The first selec
tion criteria we used was selectivity, which we
measured by analyzing oxytetracycline and tetra cycline (an impurity in oxytetra cycline form ula
tions) and determining the USP resolution and
selectivity
(a)
between the two compo unds. The
Allure?' Biphenyl and Allure?' PFP Propyl sta
tionary phases showed th e best performance
among the columns we tested (Table 1 and Figure
1).
These results suggest that the Allure?" Biphenyl
2006 vol, 1
Figure 1
Tetracycline and oxytetracycline have low pKa values that
make the analysis a challenge .
1
Peak List
Ret. Time (min.)
1.
oxytetracycline
4.92
*
degradation peak
5.66
2. tetracycline
7.10
Column:
Allure~
Biphenyl
Cal. # :
9166565
Dimensions:
150
x
4.6
mm
Particle size:
5/l~
Pore size:
60A
lCPH0342
Peak List
Ret. Time(min.)
1.
oxytetracycline
5.94
*
degradation peak
7.08
2. tetracycline
8.57
Column:
Allure~
PFP Propyl
Cal.
#:
9169565
Dimensions:
150
x
4.6
mm
Particle size:
5/l~
Pore size:
60A
Sample:
Inj.:
Cone.:
Samp lediluent:
Conditions:
Mobile phase:
Flow:
Temp.:
Del.:
20/lL
(Allure~
Biphenyl column)or 30/lL(Allure" PFP Propyl column)
100/lg/ mL
eachcomponent
methanol
20mMammoniumphosphate (pH 2.5):acetonitrile,80:20
ImL/min.
ambient
UV @ 254
nm
Table 1
Among tested columns, Allure' Biphenyl and Allure' PFP
Propyl columns show the best combination of resolution and
selectivity for tetracycl ine and oxytetracycline.
Stationary
USP
Selectivity
Phase
Resolution
(a)
Allure"
Biphenyl
5.28
1.61
Allure" PFP Propyl
4.49
1.59
Ultra
CI8
3.31
1.50
Allure~
Basix
NA
1.34
Ultra C8
NA
0.47
Ultra PFP
NA
NA
• 12 •
Website :
www.chromtech.net.auE-mail :
info@chromatech.net.auTelNo : 03 9762 2034 . . . in AUSTRALIA











